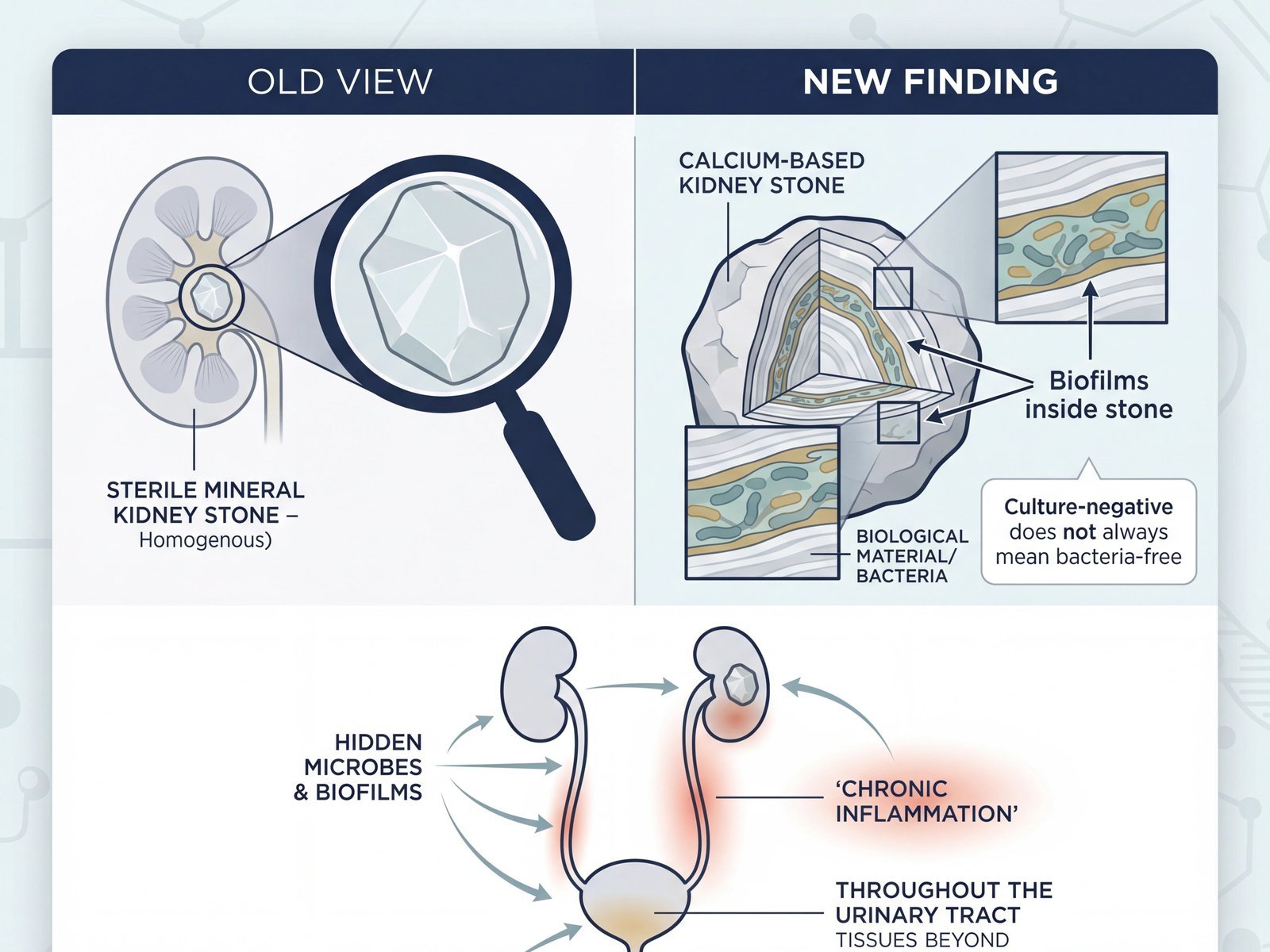
For years, patients have been told that many urinary conditions are simply “noninfectious,” meaning bacteria are not thought to play a meaningful role. A major new 2026 PNAS paper challenges that assumption in a striking way.
Researchers examining calcium-based kidney stones found that bacterial biofilms were not merely sitting on the outside as random contaminants. They were embedded inside the stones themselves, intercalated between mineral layers, even in stones that had been clinically labeled “noninfectious” and even in patients without known urinary tract infections.
What the researchers found
The study showed that calcium oxalate stones, which make up the majority of kidney stones, can contain integrated bacterial biofilms as part of their internal structure. The authors also found that areas near these biofilms had smaller crystal grain sizes, suggesting the bacteria may be helping create conditions that favor stone formation.
In plain English: the bacteria may not be innocent bystanders. They may be part of the architecture of the stone itself.
Why this matters beyond kidney stones
This matters for a much bigger reason than stones alone. It adds weight to a broader idea that standard clinical labels may be missing low-grade, hidden, structurally protected microbial processes throughout the urinary tract.
If bacteria can be built into the internal structure of a urinary stone that was considered “noninfectious,” then the old sterile-versus-infectious binary starts to look far too simplistic. “Culture-negative” does not necessarily mean bacteria-free. It may simply mean the microbes are hidden, protected, or not being detected by routine methods.
Why this is supportive of the ASU hypothesis
This new paper does not prove the entire Ascending Subclinical Urethritis hypothesis by itself. But it strongly supports several of its most important underlying ideas:
- Hidden microbial communities can exist in urinary disease even when the clinical picture does not look like a classic infection.
- Biofilms can be intrinsic to pathology rather than superficial contamination.
- Low-grade microbial persistence may help sustain chronic inflammation over long periods of time.
- Urinary tract disease may involve far more occult microbial biology than conventional thinking has allowed.
That matters because the ASU framework proposes that chronic or recurrent subclinical urethral inflammation can silently propagate upward, maintain chronic prostatic inflammation, and over time contribute to the tissue remodeling and growth we recognize clinically as BPH.
If hidden bacteria and biofilms can persist inside stones in patients considered “noninfectious,” then it becomes more biologically plausible that other urinary tract conditions may also involve persistent, underdetected microbial-inflammatory processes.
The bigger implication
One of the biggest obstacles in rethinking BPH has been the tendency to assume that if a patient does not have an obvious symptomatic infection, microbes are probably not central to the story. This new study pushes back against that mindset.
It suggests that urinary disease can include embedded microbial reservoirs that standard classifications fail to capture. That does not automatically prove an ascending urethral origin in every case. But it does make it much harder to dismiss the possibility that chronic, hidden microbial activity may be driving inflammation in ways medicine has underestimated.
And once chronic inflammation is present, the downstream consequences become easier to understand: repeated tissue injury, immune activation, remodeling, fibrosis, and abnormal growth over time. That accumulation model is highly consistent with the central logic of ASU: more time, more inflammatory events, more damage, more growth.
What this study does not prove
We should also be precise. This paper is about kidney stones, not BPH. It does not prove that every case of BPH is caused by ascending subclinical urethritis. It does not prove a specific prevention strategy. And it does not, by itself, establish that all “noninfectious” urinary diseases share the same mechanism.
But it does strengthen the broader biological foundation beneath that line of thinking: hidden bacteria, protected within biofilms and missed by ordinary clinical assumptions, may be more central to urinary pathology than many have realized.
Bottom line
This new PNAS study is important because it shows that bacteria can be woven directly into the internal structure of urinary pathology previously considered noninfectious. That is a serious conceptual shift.
For those following the emerging case for Ascending Subclinical Urethritis as an upstream driver of chronic prostate inflammation, this does not close the case. But it clearly moves the scientific conversation in the same direction: away from simplistic sterile models and toward a deeper recognition that hidden microbial processes may be shaping urinary disease far more often than we think.